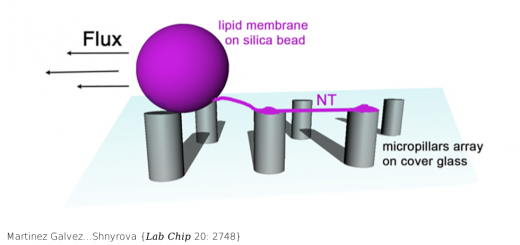
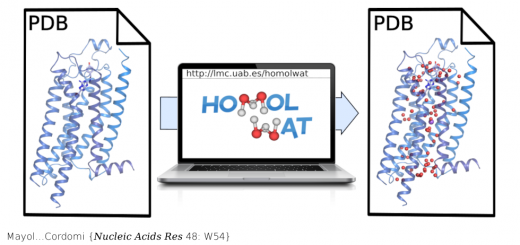
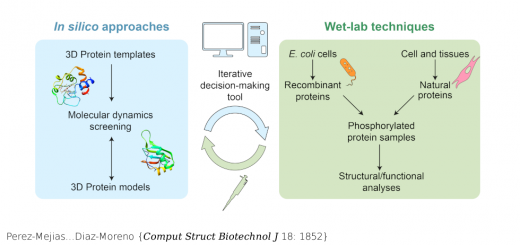
Shapes, Dynamics, and Stability of beta-Ionone and Its Two Mutants Evidenced by High-Resolution Spectroscopy in the Gas Phase

The biological unit of retinal, β-ionone, together with two mutants (a-ionone and β-damascone) have been analysed by rotational spectroscopy technique and quantum chemical calculations. β-ionone shows the flattest conformational landscape, giving it the ability to span more easily the widest variety of torsions with respect to its mutants. This feature makes it the most suitable to be part of the retinal ligand.
Uriarte I, Melandri S, Maris A, Calabrese C, Cocinero EJ.
J Phys Chem Lett 2018 Apr; 9: 1497.
The conformational landscapes of beta-ionone and two mutants (alpha-ionone and beta-damascone) have been analyzed by means of state-of-the-art rotational spectroscopy and quantum-chemical calculations. The experiments performed at high resolution and sensitivity have provided a deep insight into their conformational spaces, assigning more than 8000 transitions corresponding to the rotational structures of 54 different species (3 isomers, 14 conformers, and 40 isotopologues). Methyl internal rotation dynamics were also observed and analyzed. The work proved the great flexibility of beta-ionone due to its flatter potential energy surface. This feature confers on beta-ionone a wider ability to interconvert between conformers with rather similar energies with respect to its mutants, allowing the retinal ligand to better adapt inside the binding pocket.
PubMed: 29510049. Doi: 10.1021/acs.jpclett.8b00256.