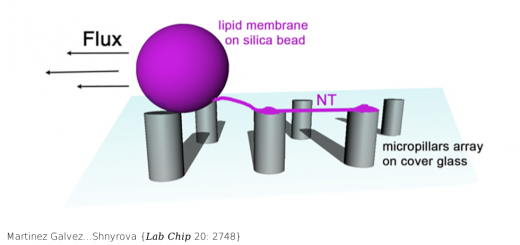
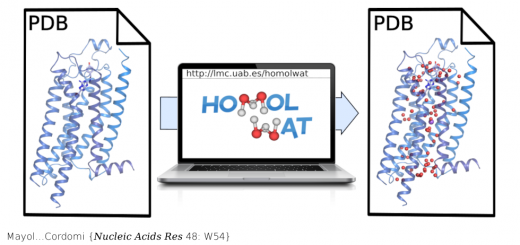
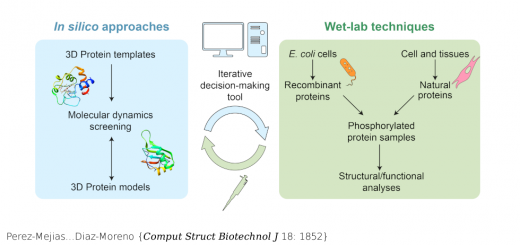
Collepardo-Guevara…Orozco {JACS 137: 10205}

The presence of acetylated lysine, one of the best known epigenetic marks, induces compaction and rigidification of histone tails in nucleosomes, thereby limiting the extend of internucleosomal interactions and triggering chromatin unfolding.
Chromatin Unfolding by Epigenetic Modifications Explained by Dramatic Impairment of Internucleosome Interactions: A Multiscale Computational Study
Collepardo-Guevara R, Portella G, Vendruscolo M, Frenkel D, Schlick T, Orozco M.
J Am Chem Soc. 2015 Aug; 137: 10205 [Epub 10 Aug 2015].
Histone tails and their epigenetic modifications play crucial roles in gene expression regulation by altering the architecture of chromatin. However, the structural mechanisms by which histone tails influence the interconversion between active and inactive chromatin remain unknown. Given the technical challenges in obtaining detailed experimental characterizations of the structure of chromatin, multiscale computations offer a promising alternative to model the effect of histone tails on chromatin folding. Here we combine multimicrosecond atomistic molecular dynamics simulations of dinucleosomes and histone tails in explicit solvent and ions, performed with three different state-of-the-art force fields and validated by experimental NMR measurements, with coarse-grained Monte Carlo simulations of 24-nucleosome arrays to describe the conformational landscape of histone tails, their roles in chromatin compaction, and the impact of lysine acetylation, a widespread epigenetic change, on both. We find that while the wild-type tails are highly flexible and disordered, the dramatic increase of secondary-structure order by lysine acetylation unfolds chromatin by decreasing tail availability for crucial fiber-compacting internucleosome interactions. This molecular level description of the effect of histone tails and their charge modifications on chromatin folding explains the sequence sensitivity and underscores the delicate connection between local and global structural and functional effects. Our approach also opens new avenues for multiscale processes of biomolecular complexes.
PubMed: 26192632. Doi: 10.1021/jacs.5b04086