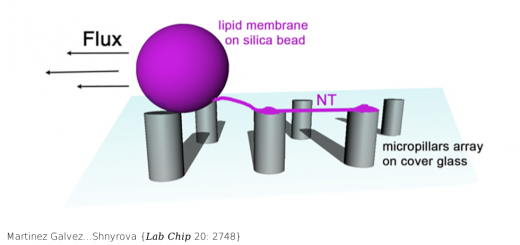
Fernández-Rivero…Prado {Sci Rep 6: 32114}

A. Nucleoplasmin (NP) binds core histones with high affinity forming complexes that differ in the stoichiometry and architecture. Binding models, based on EM data, for the interaction of NP with H2A-H2B (red) and H3-H4 (green). B. The affinity of NP for nucleosomal and linker histones is modulated by phosphorylation and intramolecular interactions that regulate the accessibility of the acidic tract at the intrinsically disordered distal face of the chaperone. Kd values for the interaction of phosphorylated (egg NP and oocyte NP) and non-phosphorylated (recombinant, full-length –rNP- or deletion mutants that lack the entire -rNPΔ120-200- or part - rNPΔ150-200- of the tail domain) NPs with histones.
A Quantitative Characterization of Nucleoplasmin/Histone Complexes Reveals Chaperone Versatility
Fernández-Rivero N, Franco A, Velázquez-Campoy A, Alonso E, Muga A, Prado A.
Sci Rep 2016 Aug.; 6: 32114.
Nucleoplasmin (NP) is an abundant histone chaperone in vertebrate oocytes and embryos involved in storing and releasing maternal histones to establish and maintain the zygotic epigenome. NP has been considered a H2A-H2B histone chaperone, and recently it has been shown that it can also interact with H3-H4. However, its interaction with different types of histones has not been quantitatively studied so far. We show here that NP binds H2A-H2B, H3-H4 and linker histones with Kd values in the subnanomolar range, forming different complexes. Post-translational modifications of NP regulate exposure of the polyGlu tract at the disordered distal face of the protein and induce an increase in chaperone affinity for all histones. The relative affinity of NP for H2A-H2B and linker histones and the fact that they interact with the distal face of the chaperone could explain their competition for chaperone binding, a relevant process in NP-mediated sperm chromatin remodelling during fertilization. Our data show that NP binds H3-H4 tetramers in a nucleosomal conformation and dimers, transferring them to DNA to form disomes and tetrasomes. This finding might be relevant to elucidate the role of NP in chromatin disassembly and assembly during replication and transcription.
PubMed: 27558753. Doi: 10.1038/srep32114.