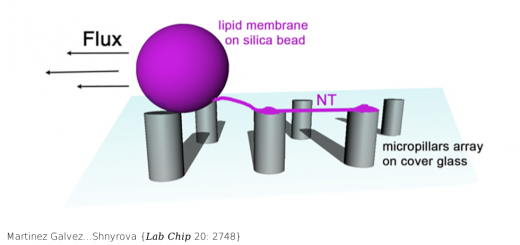
Cryo-EM structure of the adenosine A2A receptor coupled to an engineered heterotrimeric G protein
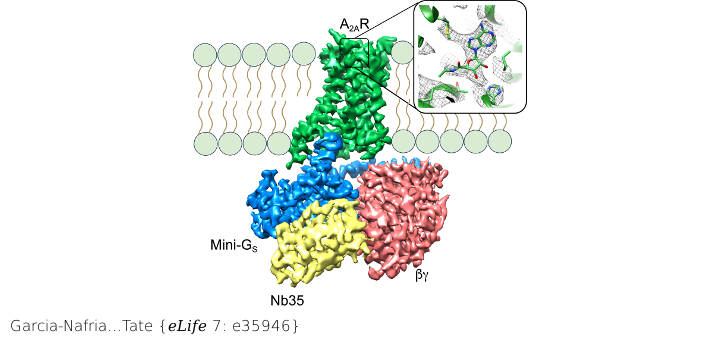
Cryo-EM structure of the human adenosine A2A receptor (A2AR) bound to the agonist NECA (zoom panel) and coupled to a engineered heterotrimeric GSβγ complex.
Garcia-Nafria J, Lee Y, Bai X, Carpenter B, Tate CG.
eLife 2018 May; 7: e35946.
The adenosine A2A receptor (A2AR) is a prototypical G protein-coupled receptor (GPCR) that couples to the heterotrimeric G protein GS. Here, we determine the structure by electron cryo-microscopy (cryo-EM) of A2AR at pH 7.5 bound to the small molecule agonist NECA and coupled to an engineered heterotrimeric G protein, which contains mini-GS, the betagamma subunits and nanobody Nb35. Most regions of the complex have a resolution of ~3.8 A or better. Comparison with the 3.4 A resolution crystal structure shows that the receptor and mini-GS are virtually identical and that the density of the side chains and ligand are of comparable quality. However, the cryo-EM density map also indicates regions that are flexible in comparison to the crystal structures, which unexpectedly includes regions in the ligand binding pocket. In addition, an interaction between intracellular loop 1 of the receptor and the beta subunit of the G protein was observed.
PubMed: 29726815. Doi: 10.7554/eLife.35946. Free PMC article