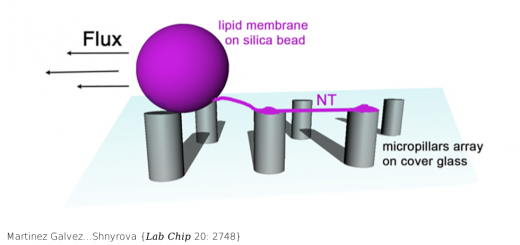
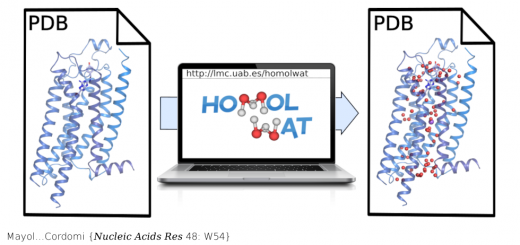
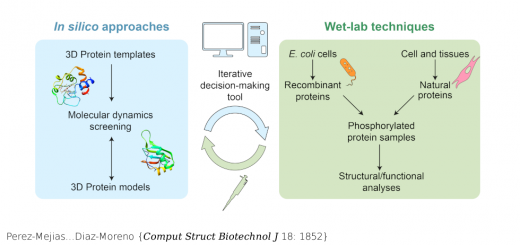
Camunas-Soler…Ritort {Nucleic Acids Res 43: 2767}

The binding sites of the anticancer drug Thiocoraline onto DNA are detected with one base pair resolution by unzipping a single DNA molecule with optical tweezers, and looking at the mechanical footprint of binding (force-extension curve in red).
Single-molecule kinetics and footprinting of DNA bis-intercalation: the paradigmatic case of Thiocoraline
Camunas-Soler J, Manosas M, Frutos S, Tulla-Puche J, Albericio F, Ritort F.
Nucleic Acids Res. 2015 March; 43:2767. [Epub 17 February 2015]
DNA bis-intercalators are widely used in molecular biology with applications ranging from DNA imaging to anticancer pharmacology. Two fundamental aspects of these ligands are the lifetime of the bis-intercalated complexes and their sequence selectivity. Here, we perform single-molecule optical tweezers experiments with the peptide Thiocoraline showing, for the first time, that bis-intercalation is driven by a very slow off-rate that steeply decreases with applied force. This feature reveals the existence of a long-lived (minutes) mono-intercalated intermediate that contributes to the extremely long lifetime of the complex (hours). We further exploit this particularly slow kinetics to determine the thermodynamics of binding and persistence length of bis-intercalated DNA for a given fraction of bound ligand, a measurement inaccessible in previous studies of faster intercalating agents. We also develop a novel single-molecule footprinting technique based on DNA unzipping and determine the preferred binding sites of Thiocoraline with one base-pair resolution. This fast and radiolabelling-free footprinting technique provides direct access to the binding sites of small ligands to nucleic acids without the need of cleavage agents. Overall, our results provide new insights into the binding pathway of bis-intercalators and the reported selectivity might be of relevance for this and other anticancer drugs interfering with DNA replication and transcription in carcinogenic cell lines.
PubMed: 25690887. Doi: 10.1093/nar/gkv087
PubMed: 25690887. Doi: 10.1093/nar/gkv087