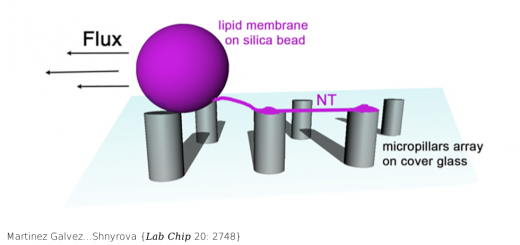
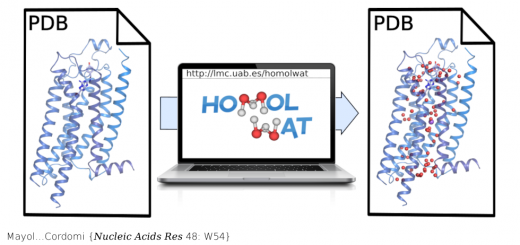
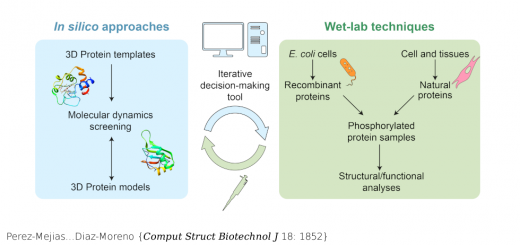
Alvira……Valpuesta {Nat Commun 5: 5484}

Structural characterization of the substrate transfer mechanism in Hsp70/Hsp90 folding machinery mediated by Hop
Alvira S, Cuéllar J, Röhl A, Yamamoto S, Itoh H, Alfonso C, Rivas G, Buchner J, Valpuesta JM.
Nat Commun. 2014 Nov 19; 5: 5484.
In eukarya, chaperones Hsp70 and Hsp90 act coordinately in the folding and maturation of a range of key proteins with the help of several co-chaperones, especially Hop. Although biochemical data define the Hop-mediated Hsp70-Hsp90 substrate transfer mechanism, the intrinsic flexibility of these proteins and the dynamic nature of their complexes have limited the structural studies of this mechanism. Here we generate several complexes in the Hsp70/Hsp90 folding pathway (Hsp90:Hop, Hsp90:Hop:Hsp70 and Hsp90:Hop:Hsp70 with a fragment of the client protein glucocorticoid receptor (GR-LBD)), and determine their 3D structure using electron microscopy techniques [….]
PubMed: 25407331. Doi: 10.1038/ncomms6484
PubMed: 25407331. Doi: 10.1038/ncomms6484