Dynamic competition for hexon binding between core protein VII and lytic protein VI promotes adenovirus maturation and entry
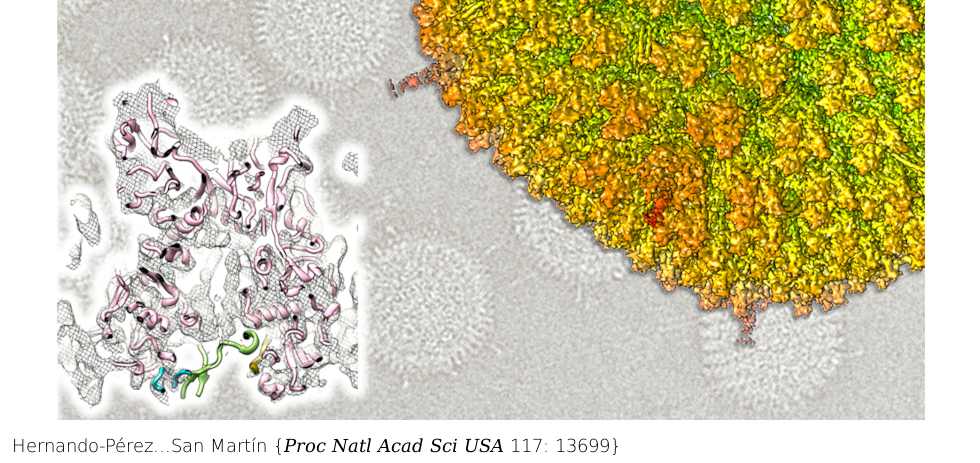
Combination of thousands of cryo-electron microscopy images (background) yields a highly detailed 3D map (top right) of human adenovirus. At the left, the structure of the major coat protein is shown fitted to the map, with empty density corresponding to the trapped lytic peptide.
Hernando-Perez M, Martin-Gonzalez N, Perez-Illana M, Suomalainen M, Condezo GN, Ostapchuk P, Gallardo J, Menendez M, Greber UF, Hearing P, de Pablo PJ, San Martin C.
Proc Natl Acad Sci U S A 2020 Jun; 117: 13699.
Adenovirus minor coat protein VI contains a membrane-disrupting peptide that is inactive when VI is bound to hexon trimers. Protein VI must be released during entry to ensure endosome escape. Hexon:VI stoichiometry has been uncertain, and only fragments of VI have been identified in the virion structure. Recent findings suggest an unexpected relationship between VI and the major core protein, VII. According to the high-resolution structure of the mature virion, VI and VII may compete for the same binding site in hexon; and noninfectious human adenovirus type 5 particles assembled in the absence of VII (Ad5-VII-) are deficient in proteolytic maturation of protein VI and endosome escape. Here we show that Ad5-VII- particles are trapped in the endosome because they fail to increase VI exposure during entry. This failure was not due to increased particle stability, because capsid disruption happened at lower thermal or mechanical stress in Ad5-VII- compared to wild-type (Ad5-wt) particles. Cryoelectron microscopy difference maps indicated that VII can occupy the same binding pocket as VI in all hexon monomers, strongly arguing for binding competition. In the Ad5-VII- map, density corresponding to the immature amino-terminal region of VI indicates that in the absence of VII the lytic peptide is trapped inside the hexon cavity, and clarifies the hexon:VI stoichiometry conundrum. We propose a model where dynamic competition between proteins VI and VII for hexon binding facilitates the complete maturation of VI, and is responsible for releasing the lytic protein from the hexon cavity during entry and stepwise uncoating.
PubMed: 32467158. Doi: 10.1073/pnas.1920896117. OPEN Free PMC
